
In the world of chemistry and manufacturing, efficiency is everything. Whether you are purifying a synthetic compound in a lab or extracting essential oils for a side hustle, knowing your percent recovery tells you exactly how much material you “saved” versus how much you lost during the process.
It is the ultimate “report card” for your technical skills. High recovery means your technique is spot on; low recovery means it’s time to check your filters and glassware for trapped product.
The Master Formula
To calculate percent recovery, you only need two numbers: what you started with and what you ended with after purification (like recrystallization).
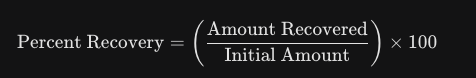
Quick Comparison: Recovery vs. Yield
Many students confuse “Percent Recovery” with “Percent Yield.” Here is the quick breakdown:
| Feature | Percent Recovery | Percent Yield |
| Goal | Measures efficiency of purification. | Measures efficiency of a chemical reaction. |
| Starting Material | An impure version of the same substance. | Different “reactant” molecules. |
| Perfect Score | 100% (No material lost). | 100% (Every molecule reacted perfectly). |
| Common Use | Recrystallization, Distillation. | Synthesis, Industrial manufacturing. |
Frequently Asked Questions
Can percent recovery be over 100%? In theory, no. In practice, yes—but it usually means your sample is still wet or contains impurities (like leftover solvent or filter paper fibers). Always dry your product thoroughly!
Why is my recovery always low? Common culprits include transferring material between too many containers, using too much solvent during washing, or premature crystallization in the funnel.
What is a “good” percent recovery? This depends on the procedure. For a difficult recrystallization, 60–70% might be excellent. For simple filtrations, you should aim for 90%+.